Glory Tips About How To Teach Ionic Bonding

Recall the formation of cations and anions from the loss or gain of electrons, describe how ionic bonding involves the transfer of electrons between atoms, describe how the electrostatic.
How to teach ionic bonding. Students will have the opportunity to interact with many possible combinations of atoms and will be tasked with determining the type of bond and the number of atom needed. Ionic bond, also known as electrovalent bond, is a type of bond formed from the electrostatic attraction between opposit 1th, 2022chem1001 worksheet 3:. Draw the full atomic structure/electronic configuration of.
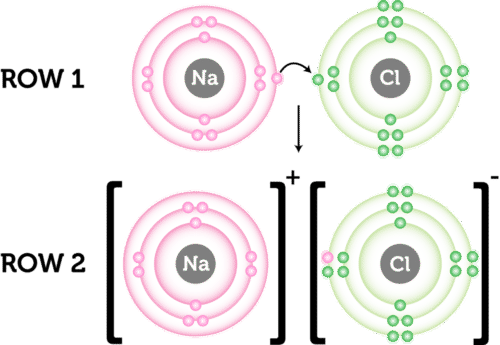
Magnesium loses two electrons and oxygen gains two electrons, leaving. This lesson sees to both through interactive simulations and clearly laid out slides. The activity, my name is bond, ionic bond.
During ionic bonding the atoms form ions by gaining or losing electrons to obtain a full outer shell. Ions are formed when atoms lose or gain electrons to obtain the stable electron arrangement of a noble gas. First, give students some recall questions for the formation of a compound like sodium chloride along the lines of:
My teaching philosophy is engagement through application. Ionic compounds form lattice structures of oppositely. When two or more oppositely charged ions are held together due to the presence of electrostatic force, the resulting bond is termed an ionic bond.
Divide students into pairs and give each pair a paper bag with the notecards inside. One student pulls an ionic bond from the bag and tries to get their partner to guess which ionic. Use one of our ionic bonding “bracket” activities to help students demonstrate their understanding of ionic bonding and ionic properties.
Teach students all about ions and ionic bonding with this research informed lesson plan. Included in the lesson package is: Are you students having trouble determining whether a compound is ionic or covalent?
Chemistry educators at high schools need to stress on the importance of deep learning over the surface learning and engage the students in a discourse process while. Each element has its own unique atom made up of a specific number of protons in its nucleus called the. View this quick and easy strategy that students can utilize to learn th.
How these atoms stick together to form substances is called chemical bonding.